Company Profile
About AIM

AIM Vaccine Co., Ltd. (Stock Code: 06660.HK) is a Hong Kong-listed company established on November 9, 2011, with a unified social credit code of 91210100583868927X and a total share capital of 1.211 billion RMB. The company's registered address is Room 412, 4th Floor, Building 6, No. 105, Jinghai 3rd Road, Beijing Economic and Technological Development Zone, Beijing.
As one of China's leading vaccine companies, AIM Vaccine covers the entire value chain from R&D, manufacturing, to commercialization. Committed to producing conscientious vaccines for the health of all, it is the second-largest and the largest private full-industrial-chain vaccine group in China, as well as a leading enterprise in the Chinese vaccine sector, with a mature and self-innovated mRNA technology platform.
AIM Vaccine owns four fully controlled licensed vaccine manufacturing companies, three vaccine research institutes, and four R&D centers, with seven R&D teams ensuring milestone delivery for its product pipeline. It is one of only two Chinese human vaccine companies with P3 laboratory strategic resources. AIM is also the largest global manufacturer of hepatitis B vaccines and the second-largest global rabies vaccine manufacturer.
The company has five validated vaccine technology platforms, offering eight commercialized vaccines and 20 vaccines under development. Its pipeline covers the top 10 global vaccine products. AIM’s commercialized products maintain market leadership and are sold across all 31 provinces, cities, and autonomous regions in China, reaching over 2,800 district and county CDCs. AIM Vaccine is a rare, comprehensive platform with advantages in pipeline, R&D, manufacturing, and sales.
Core Products Currently on Sale
Several products currently under development will be launched soon
- Serum-free rabies vaccine
- 13-valent pneumococcal vaccine (PCV13)
- 23-valent Pneumococcal Polysaccharide Vaccine (PPSV23)
- Iterative-process Highly-effective HDC Rabies Vaccine
Our Factory
- AIM Honesty
- AIM Honor
- AIM Persistence
- AIM Action

AIM Honesty
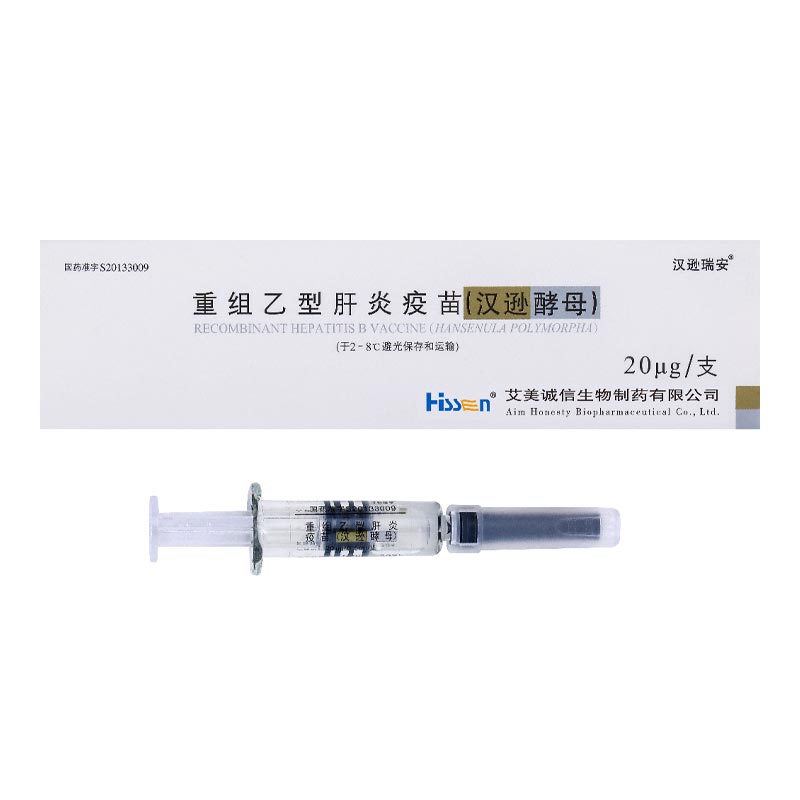
Product: recombinant hepatitis B vaccine (hansenula polymorpha)
Established in September 1993, AIM Honesty Biopharmaceutical Co.,Ltd. (AIM Honesty) is one of the earliest Chinese high-tech enterprises specializing in vaccine production, R&D and sales. With a registered capital of RMB 250 million, AIM Honesty is among the first batch of enterprise technology centers of Liaoning Province and an enterprise with national postdoctoral workstation established. Located at the beautiful coastal city Dalian—35 Wanda Road, Dalian Economy and Technology Development Area, the Company’s employees with backgrounds in medicine or related fields account for more than half of its staff.
Its main products include recombinant hepatitis B vaccine (hansenula polymorpha), etc. Hepatitis B vaccine is a national key technology research and development program of the 863 Program undertaken by AIM Honesty, with its production workshop covering over 11,000 square meters and maximum production capacity reaching 45 million doses per year. For its Hepatitis B vaccine, AIM Honesty has adopted an internationally advanced recombinant hansenula polymorpha expression technology platform, which is the first in China. Characterized by high output and high antigen presentation purity, AIM Honesty’s hepatitis B vaccine enjoys high popularity and exerts high influence in the market for its reliability, safety and suitability for a wider range of population.
At present, AIM Honesty is strengthening its production-education-research cooperation with world-renowned and domestic famous institutes, to further its development and grow bigger and stronger based on the technological superiority of its existing products. AIM Honesty is dedicated to genetic engineering vaccine platform technology
We believe, in the foreseeable future, our new vaccine products will be gradually put into the market. the production of vaccine products concerns public health and the people’s well-being. As a biological vaccine enterprise, AIM Honesty shoulders China’s public health undertaking and the social mission of healthy China. Whether in the past, at present or in the future, AIM Honesty has always been striving to become the mainstay of Chinese biological vaccine enterprises. Based on further development of existing products, we will continue to enhance the R&D of new products, continue to expand the width and depth of our product line, and at the same time work at the international forefront of biotechnology and apply modern biotechnology to produce more and better products, and make more active contribution to a win-win situation for the company and its customers, China’s cause of disease prevention and control, as well as a better life for the mankind.

AIM Honor
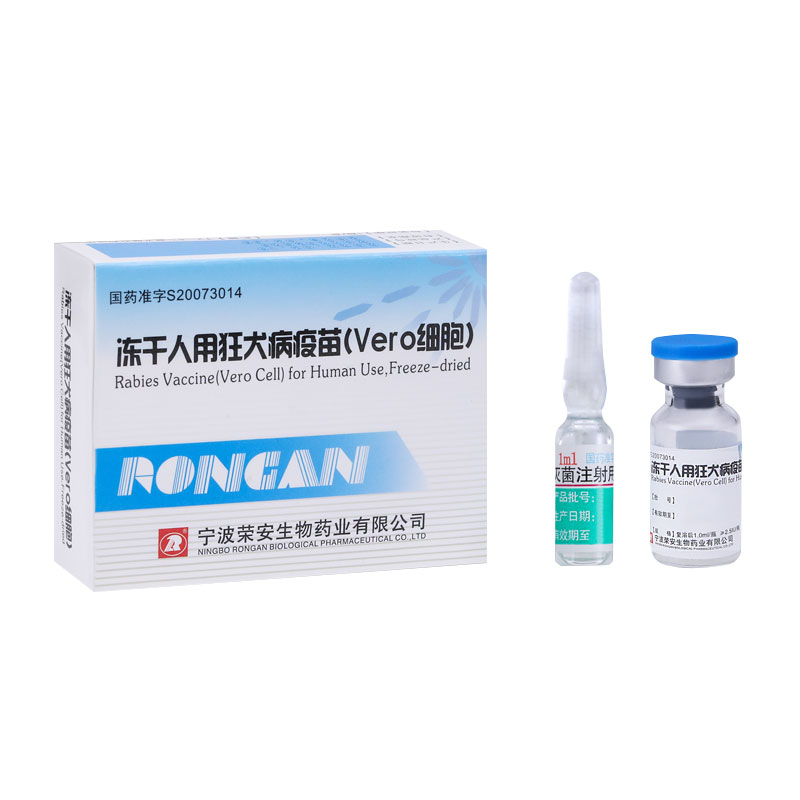
Product: rabies vaccine (Vero cell)
AIM Honor (Ningbo) Biopharmaceutical Co., Ltd. is a bio-pharmaceutical enterprise wholly owned by AIM Vaccine Co., Ltd. Established in 2001, it is a high and new technology enterprise recognized by Zhejiang Association for Science and Technology and Ningbo Association for Science and Technology.
Dedicated to the manufacturing and commercialization of rabies vaccine (Vero cell), AIM Honor has obtained the new drug certificate for rabies vaccine for human use (Vero cell) in 2001, the drug GMP certificate and drug production permit necessary for production in 2002, and the drug registration approval for freeze-dried rabies vaccine for human use (Vero cell) in September 2007. As for 2020, AIM Honor has been the second largest manufacturer of rabies vaccine for human use in China and the world.
At the present stage, AIM Honor mainly produces freeze-dried rabies vaccine for human use with an annual production of about 22 million doses. Both its product quality and market occupancy are at the industry-leading level in China. Based on its rabies vaccine for human use (Vero cell), currently AIM Honor is developing two candidate rabies vaccines for human use.
Under unified arrangement and planning by AIM Vaccine, AIM Honor is developing at a high speed. It always insists on putting product quality first and remains dedicated to the R&D of new processes and new technologies. In the future, AIM Honor will strive to become China’s top-ranking viral vaccine manufacturer by developing and applying the most advanced production technologies.

AIM Persistence
Product: live attenuated parotitis vaccine, inactivated bivalent HFRS vaccine (Vero cell), ACYW135 meningococcus polysaccharide vaccine, and HITB combined vaccine (Hib)
Since April 17, 2018, the company name “Zhejiang Weixin BioPharm (Zhejiang) Co., Ltd. “has been changed to “AIM Persistence Biopharmaceutical Co., Ltd.” Originally established in 2002, AIM Persistence Biopharmaceutical Co., Ltd. (“AIM Persistence”) is a subsidiary of AIM Vaccine Co., Ltd. It is a high and new technology enterprise specializing in the scientific research & development and production of vaccines and related biological products.
With a registered capital of RMB 515,306,120, AIM Persistence covers an area of 41263㎡ and employs 254 people, with technicians accounting for above 70% of its staff. It owns domestic first-class research & test instrument and production equipment and facilities. Since its establishment, AIM Persistence has received generous support from the Ningbo government as a recognized high and new technology enterprise significantly supported and prioritized by Ningbo. The Company has established extensive collaboration with domestic and foreign scientific research institutes and universities on projects and technology development.
At present, AIM Persistence has four major vaccine products: live attenuated parotitis vaccine, inactivated bivalent HFRS vaccine (Vero cell), ACYW135 meningococcus polysaccharide vaccine, and HITB combined vaccine (Hib). Its live attenuated parotitis vaccine obtained a permit for production in October 2004, and passed the national GMP certification in January 2005. With a total workshop area of 2340㎡ and an annual production scale of 16 million vials, this product has been sold nationwide (25 provinces and municipalities including Heilongjiang, Jilin, Liaoning, Hebei, Tianjin, Shandong, Shanxi, Shaanxi, Hubei, Hunan, Anhui, Fujian, Guangdong, Guangxi, Guizhou, Sichuan, Gansu, Qinghai, Inner Mongolia, Ningxia, Xinjiang, Yunnan, Jiangxi and Chongqing) with over 5 million vials used accumulatively. Due to its high titer level and high safety level, the live attenuated parotitis vaccine of AIM Persistence has won the trust of users, and gradually established a good brand reputation in China, with a market occupancy of above 70%. In September 2007, the inactivated bivalent HFRS vaccine (Vero cell) of AIM Persistence obtained a new drug certificate and a production permit from the National Medical Products Administration. Using WHO-approved Vero cells rather than primary animal cells, this vaccine is safe and reliable and can be mass produced with neither environmental pollution due to animal breeding nor potential exogenous hazards brought by animal cells. With the adoption of advanced production techniques and the application of modern biotechnology and purification process, it has achieved high purity, good immunogenicity, safety, effectiveness and stable quality, representing the advanced level of similar vaccines in China at present. It can be used in epidemic areas of type I or type II HFRS and mixed epidemic areas of type I & II HFRS. With a broad immunization coverage, it is nowadays the most widely used vaccine for HFRS prevention. AIM Persistence’s ACYW135 meningococcus polysaccharide vaccine obtained its new drug certificate and GMP certificate in November 2018 and was put into the market in March 2020. The group A, C, Y and W135 Neisseria meningitidis strains used by this vaccine are from the National Institutes for Food and Drug Control in compliance with the results of China’s epidemiological surveys on Neisseria meningitidis and can prevent epidemic cerebrospinal meningitis caused by group A, C, Y and W135 Neisseria meningitidis. This vaccine adopts the classic capsular polysaccharide purification process, which is the conventical preparation method for bacterial polysaccharide vaccines due to its stability and reliability. As a substitute for group A meningococcal polysaccharide vaccine (Class 1 vaccine) and group A+C meningococcal polysaccharide vaccine, AIM Persistence’s ACYW135 meningococcus polysaccharide vaccine has good market prospect.
In the future, AIM Persistence will enrich its product variety by optimizing production process, improving product quality and increasing R&D investment, to further enhance its core competitiveness. Also, it plans to introduce and attract outstanding talent to the Company through various methods and establish a corresponding talent incentive plan to maintain the enthusiasm and motivation of its staff at work. Always striving for the highest quality, with leapfrog development and care for society as its purposes, AIM Persistence Biopharmaceutical Co., Ltd. seeks cooperation based on mutual benefit and joint development, continues to optimize its industrial structure and allocation of human resources, and looks forward to establishing extensive partnerships and together creating brilliance with comrades from all sectors of the society.

AIM Action
Product: Inactivated hepatitis A vaccine
Established in 2011, AIM Action BioPharm Co., Ltd. (“AIM Action”) is a subsidiary of AIM Vaccine Co., Ltd. and one of the only two domestic manufactures of inactivated hepatitis A vaccine. With a registered capital of RMB 360 million, the Company is located at the China Medical City of Taizhou, Jiangsu Province, occupying nearly 18711㎡ of land with a total floor area of about 60000㎡。 AIM Action specializes in viral vaccine platform technologies.
In accordance with AIM Vaccine’s mission of “producing good vaccines for the health of the people,” AIM Action aims to give full play to the whole-industry-chain advantages of the group, inherit the hepatitis A production technology of the Institute of Medical Biology, Chinese Academy of Medical Sciences, and utilize the industrial cluster of China Medical City and various preferential policies, to develop biological products such as new vaccines and create a high-level industrial production platform targeting the prevention and control needs of major communicable diseases at home and abroad.
So far, AIM Action has completed the2015,struction of its inactivated hepatitis A vaccine production line, passed the national GMP certification, equipped itself with cell factory and microcarrier cell fermentation culture technologies for the mass production of biological products, and obtained a new drug certificate issued by the National Medical Products Administration on April 2, 2015 as well as a GMP certificate issued by the National Medical Products Administration for its inactivated hepatitis A vaccines (human diploid cell) of all specifications in January 2016.Looking into the future, AIM Action will promote the industrialization of its vaccines under development with the operation management mechanism of modern enterprise, integrate innovation and R&D with high-efficiency production, make new contribution to the prevention and control of major communicable diseases at home and abroad, and usher in a new era of the global cause of human health.
Product Application
prophylactic vaccination